
ECAP: The Neurocognitive Architecture of Endocannabinoid-Associated Pathways
A systems neuroscience model positioning the endocannabinoid system as a central hub within a broader molecular network of non-canonical receptors, neurotransmitters, hormonal axes, and gut-brain-immune pathways.
May 2025
The Endocannabinoid-Associated Pathways (ECAP) model offers a potentially transformative understanding of Cannabis sativa's complex effects. It moves beyond the traditional Endocannabinoid System (ECS) to map how cannabinoid signaling operates within a broader, interconnected network of molecular pathways.
Publication Status
- Format: White paper (self-published preprint)
- License: CC BY 4.0
- DOI: 10.5281/zenodo.17467525
- Citations: 100+ peer-reviewed sources spanning 1993-2023
Executive Summary
ECAP conceptualizes the traditionally defined ECS not as an isolated entity, but as a central regulatory hub deeply embedded within a larger, interconnected network of molecular pathways. The full range of cannabis's effects arises from the concerted action of its constituents on this broader network.
The framework encompasses:
- Canonical cannabinoid receptors (CB1, CB2)
- Non-canonical receptors and ion channels (TRPV1, GPR55, GPR18, GPR119, PPARs, GlyRs, 5-HT3A)
- Interacting neurotransmitter systems (dopaminergic, serotonergic, GABAergic, glutamatergic, cholinergic, noradrenergic)
- Modulated hormonal axes (HPA axis, oxytocinergic system)
- The influence of phytocannabinoids and terpenes (the "entourage effect" recontextualized mechanistically)
- The gut-brain-immune axis and inflammatory pathways
How the ECAP Model Is Organized
The paper is structured in 11 sections covering the full architecture:
Sections 1-2: Core Receptor Landscape. CB1 and CB2 receptors, their neurocognitive and physiological roles, plus the expanded non-canonical targets (TRPV1 channels for pain and sensory processing, GPR55 for neuromodulation and metabolism, PPARs, GlyRs, and voltage-gated channels).
Sections 3-4: Neurotransmitter and Hormonal Integration. How cannabinoid signaling interacts with dopaminergic pathways (reward, motivation), serotonergic pathways (mood, anxiety), GABAergic and glutamatergic balance (excitation/inhibition), the HPA axis (stress response, cortisol), and the oxytocin system (social bonding, trust).
Section 5: Creativity, Action, and Performance (CAP). A speculative but structured extension mapping how specific ECAP pathway activations may modulate neurocognitive states conducive to enhanced creativity (via dopamine, 5-HT1A, CB1/DMN modulation), optimized action (via dopamine, HPA axis, noradrenergic pathways), and peak performance (via GABA/glutamate balance, oxytocin, ACh). Explicitly labeled as requiring substantial empirical validation.
Sections 6-7: Terpenes and the Gut-Brain-Immune Axis. How terpenes (limonene, pinene, linalool, myrcene, beta-caryophyllene) act as aromatic modulators across the ECAP network. How cannabinoid influence on gut microbiota, intestinal permeability, and neuroinflammation feeds back into the system.
Sections 8-11: Applications, Research Gaps, and Conclusions. Practical use cases (chronic pain, anxiety, sleep, focus, neurocognitive recovery), clinical applications (neuropsychiatric, neurodegenerative, inflammatory, pediatric epilepsy, oncology supportive care), and a call for interdisciplinary research.
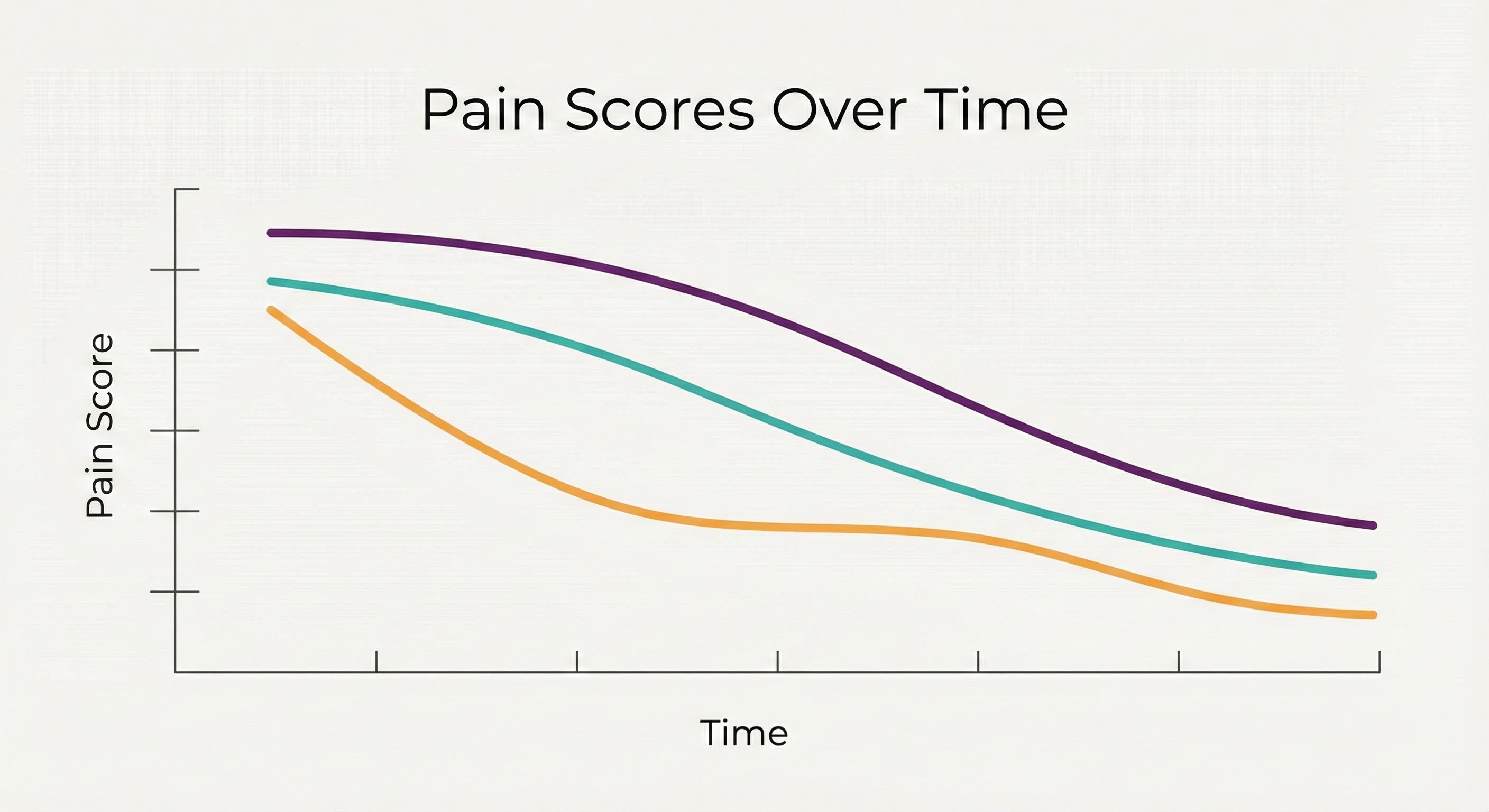
Figure A compares pain score trajectories across three ECAP-aligned intervention groups over a 12-week period.
Key Propositions
ECAP advances several testable propositions:
-
The Network Model. Cannabis effects arise from concerted action on an integrated network beyond CB1/CB2, including TRPV1, GPR55, PPARs, neurotransmitter systems, hormonal axes, and the gut-brain-immune axis.
-
Mechanistic Entourage Effect. The entourage effect is explained by parallel activations across multiple, distinct ECAP pathways converging on shared signaling molecules, neural circuits, or physiological processes.
-
Biased Agonism. Different cannabis constituents can "steer" CB1 signaling towards specific ECAP sub-pathways via functional selectivity, explaining why some experiences are anxiolytic while others are anxiogenic.
-
Chemovar Fingerprints. Specific cannabinoid/terpene ratios create unique "ECAP activation fingerprints" that produce predictable and distinct outcomes.
-
Individual Variability. The ultimate effect is an emergent property of the interaction between a product's ECAP engagement profile and the individual's pre-existing ECAP baseline conditions (genetics, gut microbiome, neurotransmitter tone, HPA axis reactivity).
-
The CAP Engine. Specific ECAP pathway activations can modulate states conducive to enhanced Creativity, optimized Action, and peak Performance. (Explicitly speculative, requiring rigorous validation.)
-
Gut-Microbiome Responsivity. Individual differences in gut microbiota composition could be a major predictor of cannabis response, and "gut typing" could become as important as pharmacogenomic testing.
Selected Figures
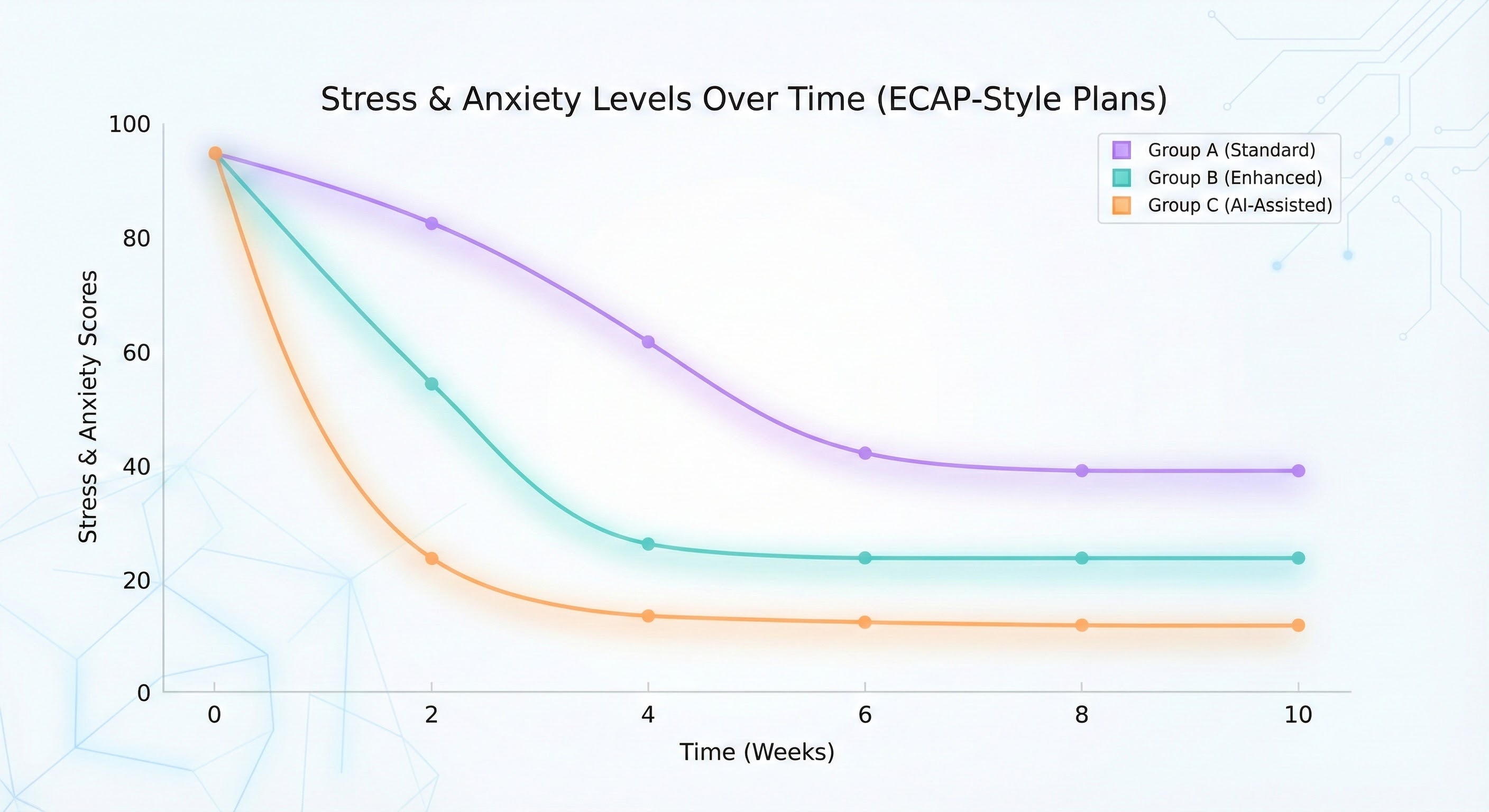
Figure B shows stress and anxiety levels across Standard, Enhanced, and AI-Assisted ECAP-aligned plans.
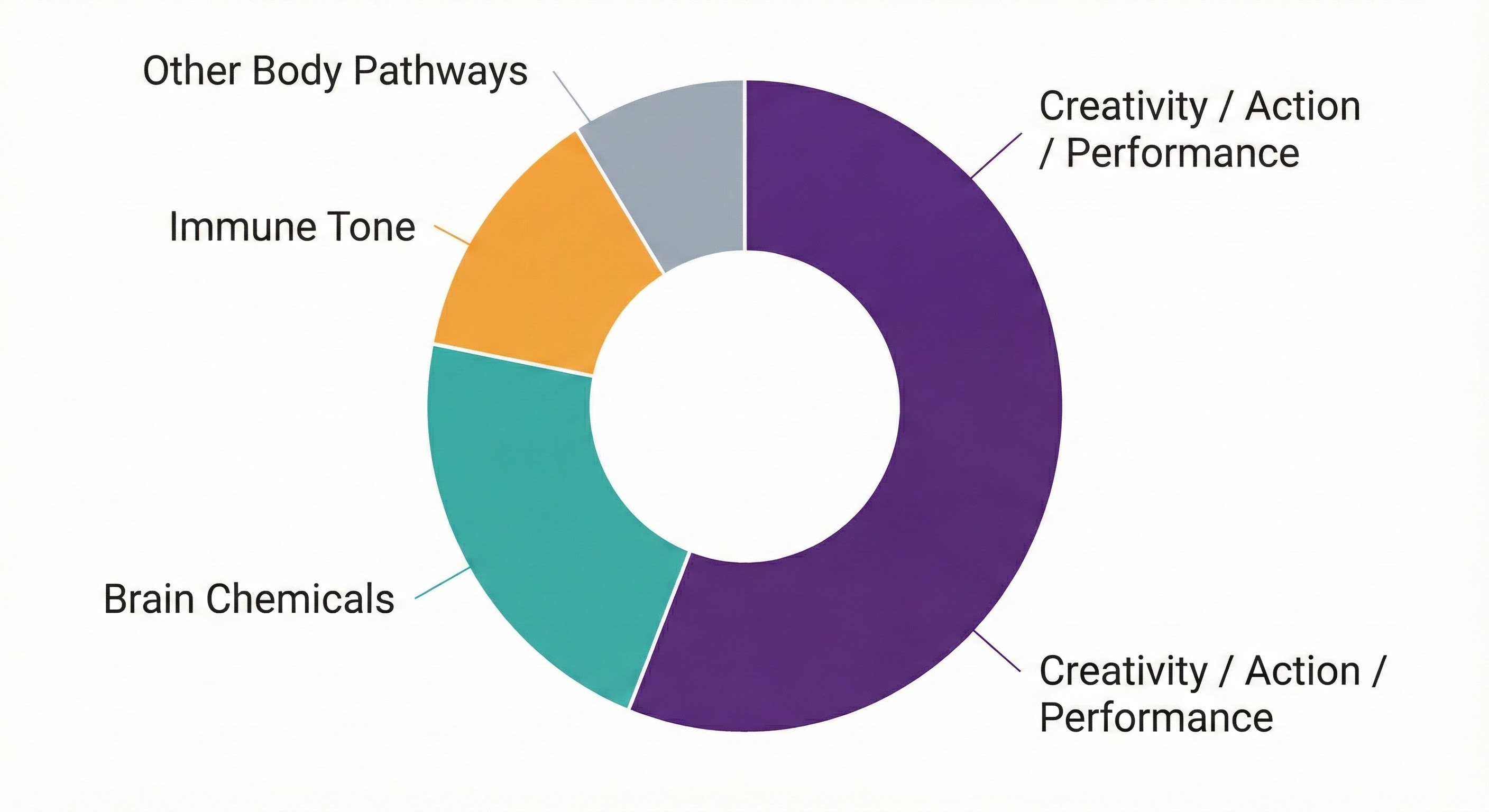
System coverage snapshot showing pathway distribution across ECAP domains. Note: the CAP (Creativity/Action/Performance) domain is a speculative extension requiring further validation.
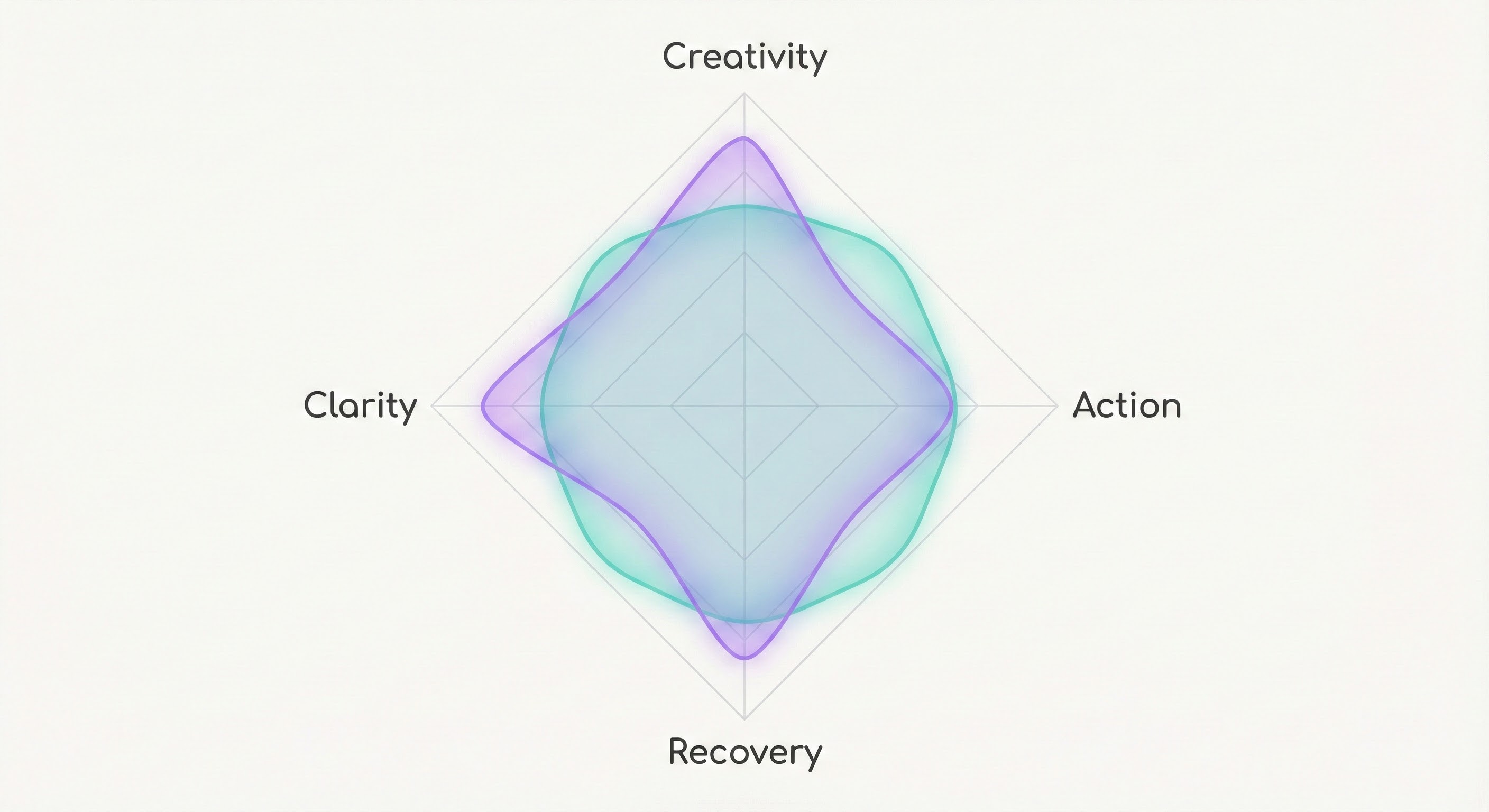
Figure C presents a radar chart comparing two hypothetical chemovar fingerprints across Creativity, Action, Recovery, and Clarity dimensions.
AI-Augmented Research Methodology
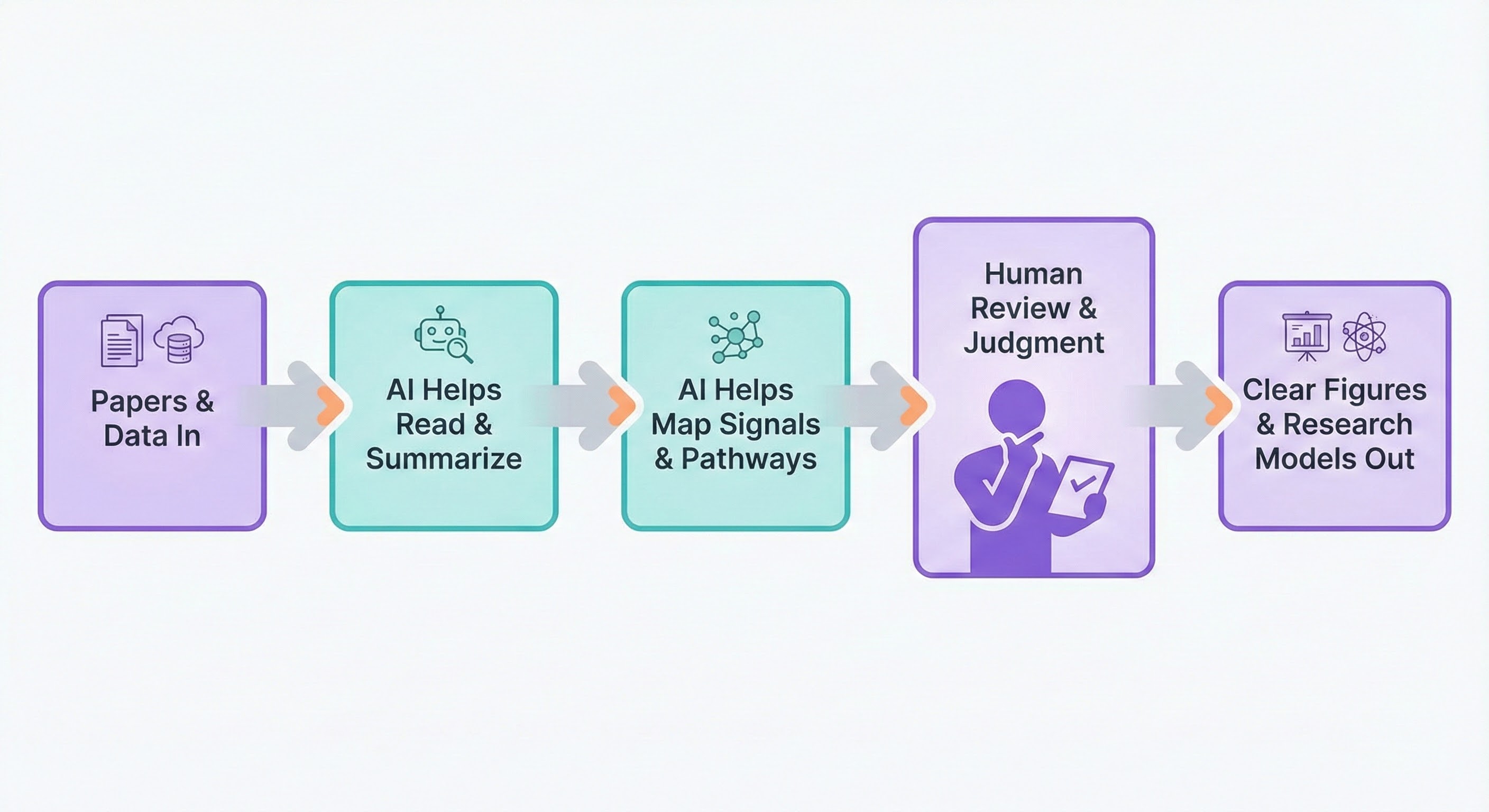
ECAP was built with AI as a research partner. AI helped organize over 100 sources, identify cross-pathway patterns, and generate initial hypothesis structures. Every connection in the model was validated against peer-reviewed literature by a licensed pharmacist with 18 years of clinical experience. AI did not replace pharmacological judgment. It accelerated the synthesis.
Who This Is For
- Cannabis brands and product teams who need science-backed formulation guidance
- Clinicians and mental health professionals who want a structured framework for understanding cannabinoid interactions
- Researchers and students looking for testable propositions and a systems-level map
What Comes Next
ECAP is the first of three interconnected research publications:
- ECAP (released) - The neurocognitive architecture of endocannabinoid-associated pathways
- The A2 System (working paper) - A unified framework mapping attention and intention onto the Expected Free Energy equation
- The Abundance Thesis (in development) - Evidence-based economic empowerment pathways
Access the Research
Download Full Paper (PDF) via Zenodo DOI: 10.5281/zenodo.17467525
Licensed under CC BY 4.0. Figures may be reused with attribution.
Selected References
- Ben-Shabat, S., et al. (1998). An entourage effect: inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. European Journal of Pharmacology, 353(1), 23-31.
- Russo, E. B. (2011). Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British Journal of Pharmacology, 163(7), 1344-1364.
- Pertwee, R. G. (2008). The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids. British Journal of Pharmacology, 153(2), 199-215.
- Zou, S., & Kumar, U. (2018). Cannabinoid receptors and the endocannabinoid system: signaling and function in the central nervous system. International Journal of Molecular Sciences, 19(3), 833.
- Gertsch, J., et al. (2008). Beta-caryophyllene is a dietary cannabinoid. Proceedings of the National Academy of Sciences, 105(26), 9099-9104.
- De Petrocellis, L., et al. (2011). Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. British Journal of Pharmacology, 163(7), 1479-1494.
- Garcia-Gutierrez, M. S., et al. (2020). Cannabidiol: A potential new alternative for the treatment of anxiety, depression, and psychotic disorders. Biomolecules, 10(11), 1575.
Author: Dr. Jeff Bullock, PharmD | ORCID: 0009-0009-2053-4854
Bibliography
- Mechoulam, R., & Parker, L. A. (2013). The endocannabinoid system and the brain. Annual Review of Psychology, 64, 21-47.
- Friston, K. (2010). The free-energy principle: a unified brain theory? Nature Reviews Neuroscience, 11(2), 127-138.
- Russo, E. B. (2011). Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British Journal of Pharmacology, 163(7), 1344-1364.
- Di Marzo, V. (2018). New approaches and challenges to targeting the endocannabinoid system. Nature Reviews Drug Discovery, 17(9), 623-639.
- Fries, P. (2015). Rhythms for cognition: Communication through coherence. Neuron, 88(1), 220-235.
Full bibliography (100+ sources) available in the complete paper.